Ion bonding table1/31/2024 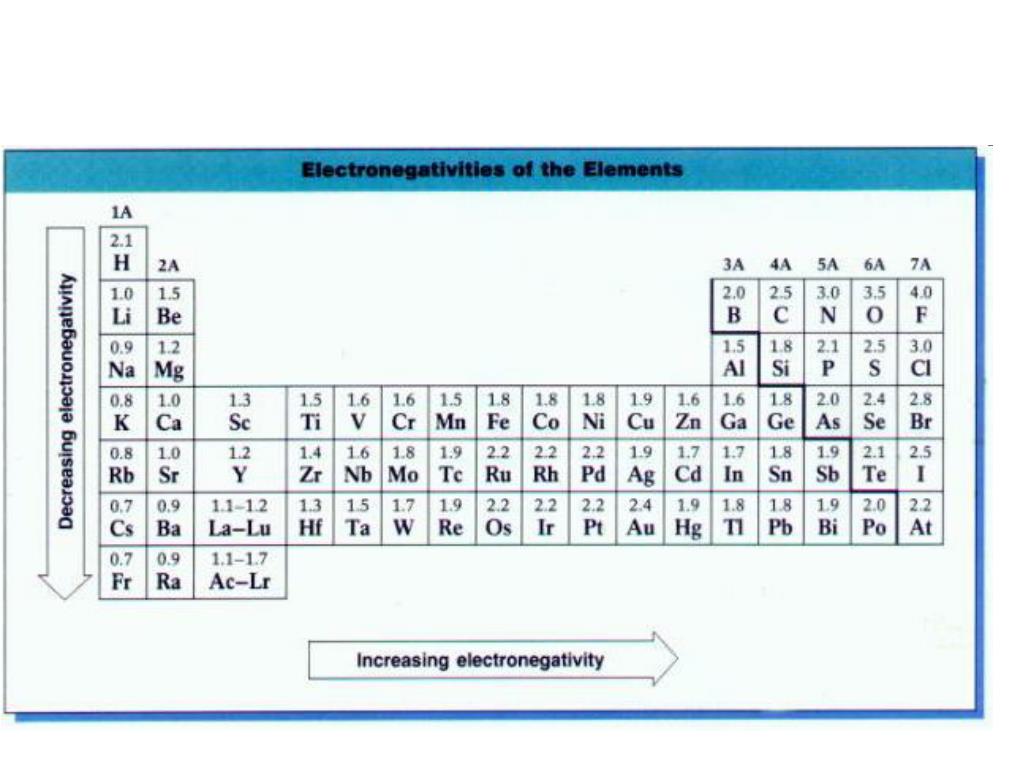
“ Dogs Teaching Chemistry - Chemical Bonds” courtesy of Lauren Girard. Ionization energy is the smallest amount of energy needed to pull an electron away from an atom in the gas state. Ions are single charged atoms (simple ions) or small charged molecules (polyatomic ions). Chemical bonds are formed when electrons in different atoms interact with each. Ion Definition Table of Ions Table of Ions An ion is a small electrically charged particle. In a carbon-oxygen bond, more electrons would be attracted to the oxygen because it is to the right of carbon in its row in the periodic table. Solutions for Lecture 9 (PDF) Acknowledgement Table salt is sodium chloride (NaCl), which is a simple compound of two. Kateryna Kozyrytska, a female biochemist born in Ukraine, describes how as a scientist she doesn’t have to choose between career and home-life like some of her peers back home, that there are examples, even at MIT, of women who have both. Kateryna Kozyrytska discusses her research on proteins that catalyze a carbon-chlorine bond-forming reaction, and she describes how taking advantage of chlorine’s electronegativity may lead to new medications to fight bacteria and the other “bugs” that make us sick. Lecture 9 Clicker Questions (PDF) Textbook ReadingĪpplying the Special Properties of Halogens in Drug Designīacteria can develop resistance to antibiotics, creating a need for current and future scientists to develop new types on antibiotics.  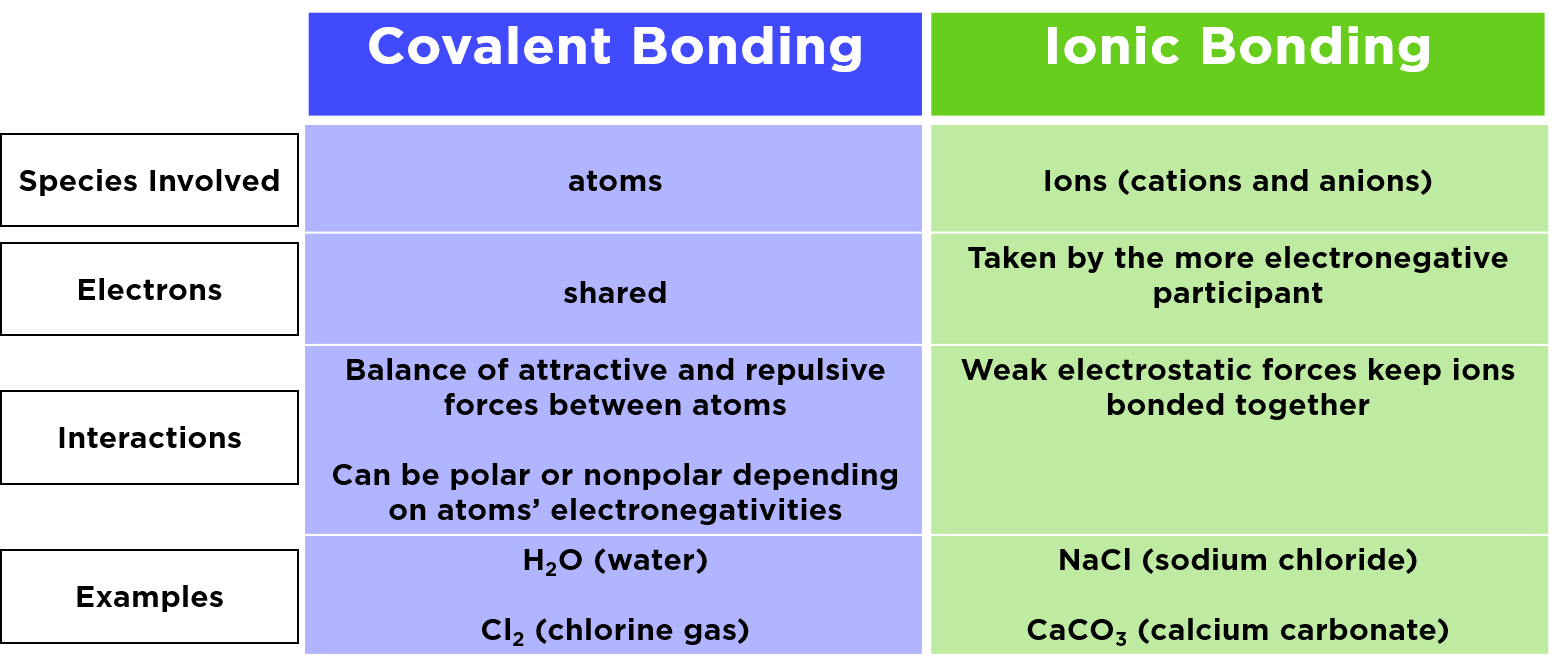
Ionic bonds occur between metals and non-metals on the periodic table. Notes for Lecture 9 (PDF) Clicker Questions In modern language, the central idea of an ionic bond is that electrons (one or.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
